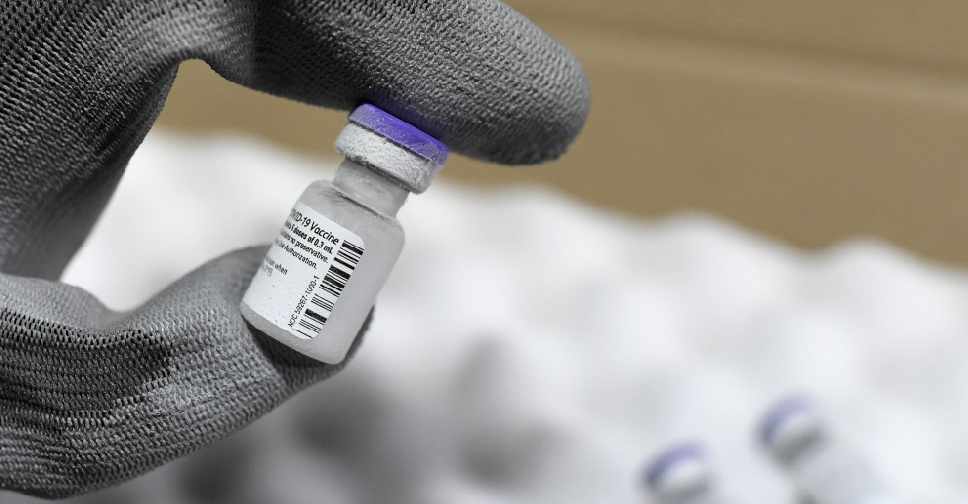
The Philippines is evaluating the emergency use of Pfizer's COVID-19 vaccine, the presidential spokesman said on Saturday.
Pfizer was the first company to seek the Philippine regulator's approval for emergency use of its coronavirus vaccine, Harry Roque, spokesman of President Rodrigo Duterte, said in a statement.
It will take the food and drugs agency 21 days to evaluate and approve the vaccine, he said, adding that inoculation would start as soon as stocks become available.
The Philippines has the second highest number of COVID-19 infections and deaths in Southeast Asia, next to Indonesia.


 Four countries condemn Iranian attack on Ras Laffan in Qatar
Four countries condemn Iranian attack on Ras Laffan in Qatar
 Netanyahu says Iran no longer has uranium enrichment capacity
Netanyahu says Iran no longer has uranium enrichment capacity
 US objectives in Iran have not changed, says Hegseth
US objectives in Iran have not changed, says Hegseth
 Trump vows no more attacks by Israel on Iran gas field
Trump vows no more attacks by Israel on Iran gas field
 Trump hails Japanese leader, says Tokyo 'really stepping up to the plate' on Iran
Trump hails Japanese leader, says Tokyo 'really stepping up to the plate' on Iran




