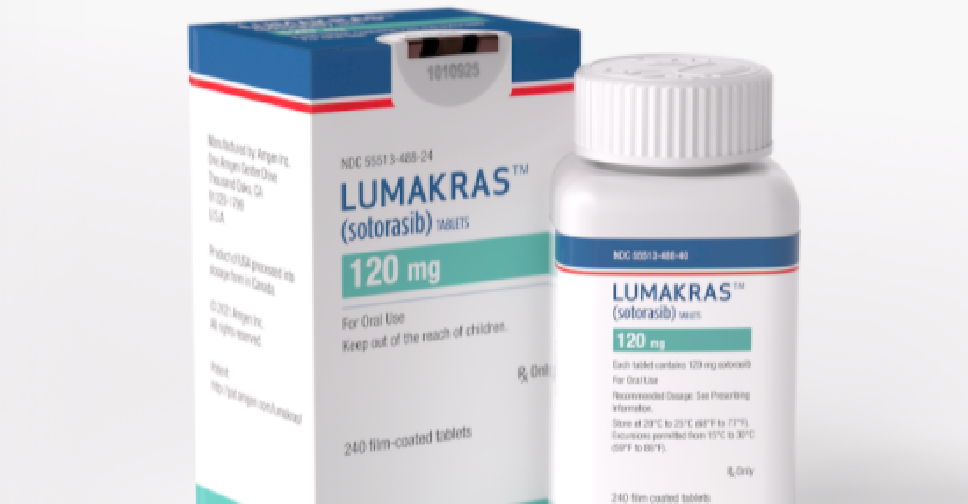
The UAE has become the second country in the world to approve the new lung cancer drug 'Lumakras'.
The Ministry of Health and Prevention (MoHAP) has cleared the registration and use of the drug, which recently received the approval of the US Food and Drug Administration (FDA).
It will help speed up the treatment plan of patients in the UAE and improve their quality of life.
Lumakras, manufactured by Amgen, is prescribed to adult patients with KRAS G12C-mutated locally advanced or metastatic non-small cell lung cancer (NSCLC), who have received at least one previous cancer therapy.
It is supplied as film-coated tablets for oral use containing 120 mg of sotorasib.


 UAE suspends new visas for Ebola-hit DRC, Uganda, South Sudan nationals
UAE suspends new visas for Ebola-hit DRC, Uganda, South Sudan nationals
 Dubai Humanitarian airlifts medical supplies to support Ebola response
Dubai Humanitarian airlifts medical supplies to support Ebola response
 Dubai Police issues over 4,500 hard shoulder violations
Dubai Police issues over 4,500 hard shoulder violations
 UAE brokers another Ukraine-Russia prisoner swap
UAE brokers another Ukraine-Russia prisoner swap
